A new era is beginning
COVID-19 has placed the healthcare sector front and centre in all our minds. The development of a vaccine in record time was truly remarkable, showing what can be achieved when great minds and innovative technology come together. Once the 'poor cousin', the biotech sector is coming into its own and is now at a key inflection point. Record funding and the entry of a 'new breed' of players, thanks to the wonders of AI and computer power, is driving considerable transformation, especially in drug discovery. What does this mean for the existing players, particularly traditional pharma? How can they compete with this new breed?
The past 18 months has shown us how a virus with a diameter of just 110 nanometres can stop the world in its tracks and impact our daily lives. I studied virology in the 1990s because I was fascinated by HIV, a virus that can integrate its genetic information into the human genome using its own enzyme. Through this integration, the virus hides from the immune system. It is a remarkable, efficient engineering process. So far, a vaccine or cure for HIV remain elusive, but fortunately, therapeutics are available. It took about two years to identify what causes AIDS in the early 1980s, and another four years to release the first drug. Biotech was in its infancy back then and pharma stepped up to the task.
This time, during the COVID-19 pandemic, biotech has taken charge, and molecular engineering and computer sciences have moved to the forefront. Progress was made in weeks vs. years. This is science at its best - and that is what the future of biotech has in store for us.
The biotech industry is all about efficiency; focusing on transforming the drug discovery process, by tapping into biology, engineering and computer sciences. These three disciplines, when working in unison, will continue to have wide-ranging ramifications for the healthcare sector and other industries over the next decade and beyond.
Challenges will arise and hesitation will persist, but if history is any guide, they will gradually be overcome, with humanity the chief beneficiary.
“I think the biggest innovations of the 21st century will be at the intersection of biology and technology. A new era is beginning.” - Steve Jobs (2011)(1)
New therapeutic modalities are successfully challenging the status quo
In the late 1970s, Genetic Engineering Technologies - Genentech, a San Francisco-based biotech, set out to commercialise genetic engineering, a technology that was rapidly being adopted in research labs. The idea of making therapeutics in bacteria or cells was frowned upon by many, as it challenged the status quo, which in this case, was the small molecule pill, chemically synthesised by machines and sold in the primary care market. Despite all the hesitation, Genentech persisted. Fast-forward to today and genomics and molecular engineering are at the core of the biotech and pharmaceutical industry. In April this year, the United States Food and Drug Administration (FDA) approved the 100th monoclonal antibody, a therapeutic modality that had its debut in 1986.

Source: Sally Smith Hughes, "Genentech – The Beginnings of Biotech”, The University of Chicago Press, April 2013.
These gentlemen were responsible for the first cloning of insulin for Genentech.
The increase in diversity of therapeutic modalities has been a key theme over the past decade, and given molecular engineering advances, more expansion is expected, helping us to pair the right modality with the ideal drug target. Most recently, despite many naysayers, messenger RNA (mRNA) was added to the therapeutic armamentarium. Again, it showed how hard work, persistence, solid funding and exceptional teams can overcome negativity and resistance.
On numerous occasions, a new approach is greeted with great hesitation, but that is what science is about - testing a hypothesis, not reinventing an already validated concept.
It is not just therapeutic modalities that have seen great advances; our understanding of the molecular profile of diseases has progressed immensely as well. We are big believers in precision medicine and are confident that neurology will similarly follow oncology’s targeted therapy pathway. Oncology once had chemotherapy as its only weapon at its disposal. Today, we can classify cancer by its molecular profile rather than its location and treat it accordingly.
Neurological diseases are complex and they take years to manifest clinical symptoms. There are genes that when mutated can cause, or put you at risk of developing, neurodegenerative diseases. In the case of Alzheimer’s disease, there are 35 known genetic associations and many are linked to the brain’s immune system, while in the case of Parkinson’s disease, there are over 95. The ability to identify and develop a targeted drug for each mutated gene is not beyond the realms of possibility in the future. We are already seeing success in this space.
In the coming decade, the speed of scientific progress will accelerate. With the standard of care for many diseases improving, disease prevention will take centre stage and biology will play a role in sectors outside of healthcare. It will not be without challenges, but nothing is in this industry. It is about embracing the unknown and carefully and factually considering “what if it works?”.
Biotech is reaching a key inflection point
There is no doubt in our minds that the biotech industry is at a key juncture in its short history. Today, the two worlds of biology and technology are colliding. Both had a key moment in the 1950s. James Watson and Francis Crick provided us with the chemical structure of our genetic code; while Claude Shannon and Alan Turing gave us computer code. As Walter Isaacson wrote in his recent book “The Code Breaker”:
“Now we have entered a third and even more momentous era, a life science revolution. Children who study digital coding will be joined by those who study genetic code.” - Walter Isaacson(2)
There is a mindset shift occurring in the biotech industry that is driven by the convergence between biotech, engineering and technology. This shift is particularly interesting in the drug development industry. Pharma is no longer the epitome of drug development; it is biotech that offers very attractive alternative business models.
Over the past 10 years, the drug discovery process has been changing, partly due to advanced drug discovery tools, but also due to a significant increase in funding. In 2020, biotech funding soared to a new record high of around US$90 billion, up c.60% from the previous annual record of US$55 billion (see Fig. 1). 2021 is also shaping up to be another strong year, with funding reaching US$54 billion for the year to date as at July (+2% year-on-year vs. 2020).(3)
Fig. 1: Biotech Funding Reached Record Highs in 2020
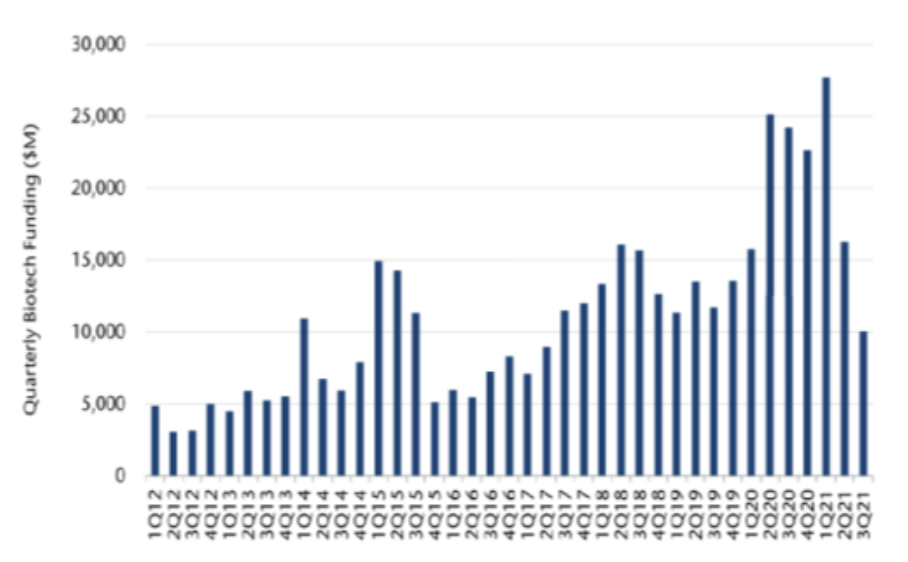
Source: FactSet Research Systems, Jefferies LLC
The funding environment for biotech companies has changed immensely. The idea that pharma companies represent one of a few funding options for biotechs is history. Today, there are several dedicated healthcare funds, various cross-over funds and tech-oriented funds that have expanded the funding universe. With the understanding that this can be a long and sometimes 'bumpy' journey, companies will often only seek those investors/funds who have a similar long-term horizon.
The calibre of management teams has also changed. Many teams are no longer novices, they have runs on the board and often know the pitfalls of large organisations. Different business models are emerging and many senior executives are leaving big pharma/biotech companies and joining biotechs.
Drug discovery is undergoing a huge transformation
The most significant change, however, is occurring in drug discovery itself. Developing new drugs is a lengthy process, starting with target and lead drug identification, moving to clinical trials and finally, commercial manufacturing. It takes years and considerable investment. This longstanding, traditional process is, however, being challenged as new discovery and drug engineering tools, automation and machine learning come together. Today, scientists, thanks to advances in computer power, can collect more data than ever before, and analysing the data is cheaper than it has ever been. Over the past 10 years, new biotechs have emerged that are taking advantage of these computer advances and challenging different stages of the drug development and manufacturing process. The aim is to make it more efficient, reduce time and ultimately save significant money.
It all starts with a specific target (e.g. a protein) and the notion that it plays a significant role in causing a disease. Initially, literature review along with experimental validation occur to confirm the importance of the target. This is followed by understanding the composition of the target better and deciphering how a drug can best interfere with the pathological activity of the target. Drug engineers will then get together and design leads that will be tested in screens to determine if they have the desired effect. The engineers also have to consider how the drug will ‘distribute’ in humans.
A lot of analysis has to occur, which is ideal for machine learning and artificial intelligence (AI). Yes, AI is a buzzword these days, but companies are really making incredible progress in drug discovery using this approach. They have focused on combining a myriad of externally sourced datasets with their own datasets generated in their laboratories. This new breed of companies employs a combination of computer and data scientists, as well as ‘drug hunters’ and 'drug engineers'. These companies have managed to bring together different skillsets and merge cultures – which is a tremendous achievement.
UK company Exscientia is one such example of this new breed of biotech company. Their AI-driven approach (see below) can save years of drug discovery, while also generating more precise leads and chemical structures that often would not have been considered.
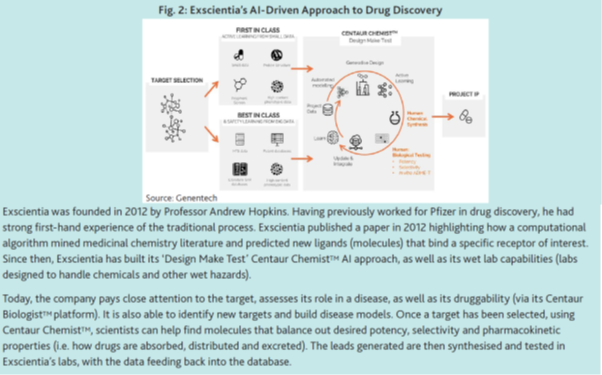
Exscientia CEO, Professor Andrew Hopkins believes computers will see molecules that traditional screens would not have been able to:
“You would never find these molecules by large-scale, high-throughput screening. It’s not looking for a needle in a haystack. It’s looking for a needle on the farm.” - Professor Andrew Hopkins, CEO, Exscientia.(4)
Exscientia is not the only company working on ‘drug discovery 2.0’. Others, like Recursion, a US-based biotech, focus on imaging cells that have been treated with drugs or harbour genetic changes. The company runs millions of experiments a week imaging the cells. It then uses machine learning to gain an insight into the cellular networks, which can translate into future treatment approaches. The naked eye is unable to see minute differences, while computers can.
To reiterate, the automation of experiments is providing the ability to collect considerably more data than ever before, with improved molecular lab tools along with computer science, making all this possible.
In the coming years, we expect the drug discovery process for small molecules and biologics will be transformed and AI will play a key role in the drug engineering and drug discovery process. It is not just drug discovery though; clinical development will also follow suit as these companies look to recruit patients and improve the design of clinical trials.
The AI drug discovery model is at an early stage; drug candidates are just starting to move into clinical development. There are signs, however, that new molecules are being developed more quickly, as is the number of quality leads, which all means faster and more cost-effective clinical development. This is very exciting, but for investors like us, the question is how will it affect the drug development landscape and the players within it?
An increasing role for new players
As always in this industry, there is deep ingrained hesitation about a ”new way of doing things”, but these new breeds of companies are not simple start-ups, they have the right ingredients, in terms of people, science and funding. Furthermore, they are making progress, gaining a foothold with biotech and pharma companies alike.
What is interesting, is that some of these next-generation drug-engineering companies are building their own drug pipelines and rapidly leapfrogging previous efforts by pharma companies. This begs the question: where does this leave the juggernaut pharma companies? For now, they have collaborated with the newcomers, making sure it does not disrupt their own internal drug discovery approach.
Some pharma companies have taken equity stakes to cement an alliance, but these biotechs will be cautious not to align themselves too closely with one partner. These ‘new’ companies have bigger ambitions and believe they can be more efficient than pharma. For pharma, the question will be how to rationalise research and development (R&D), but for now, running internal and external R&D in parallel continues.
Interestingly, some large drug developers, like Bayer, who struggled with internal R&D efficiency, have more gumption and are embracing drug discovery 2.0 a lot more.
Overall, today, there are now biotechs that resemble more efficient next-generation pharma companies. In many ways the cards are being reshuffled.
Investing in the science and the people
Returning to where we began - the efficient virus, which systematically takes advantage of the biological system and adapts over time. The next decade of drug discovery is also about systematically engineering a drug, by taking advantage of computer science and translational biology. Over time, the approach will create variations and efficiency should prevail.
To us, investing in biotechs is not about following the crowd, monitoring catalysts, quarterly earnings numbers or marginal improvement; it is about leaps in science, ongoing progress, the people behind the ideas, their unrelenting determination, independent thinking and their ability to step-up to the challenge.
My time in the pharmaceutical and biotech industry taught me that facts drive decisions and can overcome preconceived opinions and challenges. In this industry, you cannot be complacent or rely on a spreadsheet. Investing in this sector requires a deep understanding of the industry, an intense and dogged approach to research, an open mind, and importantly, a long-term perspective.
A patient approach can be very rewarding for those who are prepared to do the ground work, dial down the market noise and stay the course, as our investment in Moderna and BioNTech can attest to (see our case study).
Conclusion
As the industry evolves, the focus areas will change, but the process and thinking required to identify the next round of winners won't. Today, we have large pharma companies with some biotechs muscling in. However, in the future, there will be even more biotechs, and pharma companies will be forced to adapt and redefine themselves.
It is not just the advances in technology that are driving this incredible change, but also the funding environment for biotechs. Investors are happy to support new technology and starting a biotech is not as difficult as it once was, as the 'tools' can be outsourced.
We have the 'perfect storm'. It truly is a very exciting time to be investing in healthcare.
Learn more about the leaders of tomorrow
We seek to take advantage of the changes and developments occurring in the fields of health care and medicine, and offer investors exposure to companies engaged in the provision of a wide range of products and services within the global healthcare ecosystem.

Footnotes
(1) Source: (VIEW LINK)
(2) Walter Isaacson, "The Code Breaker Jennifer Doudna Gene Editing and the Future of the Human Race", Simon & Schuster, March 2021.
(3) Source: FactSet Research Systems and Jefferies LLC. As at July 2021.
(4) Source: (VIEW LINK)
2 topics
1 fund mentioned

